Risk factors of intra-abdominal bacterial infection after liver transplantation in patients with hepatocellular carcinoma
Introduction
Liver transplantation (LT) has achieved great development since the 1960s. As the progress of hemostatic techniques, surgical technique, and immunosuppressive drugs, operation safety has been further improved, with less postoperative complications. However, postoperative infection is still a common complication after LT, whose incidence is up to 66% (1), of which the bacteria infection accounted for about two-thirds (2). The fatality rate of postoperative infection is as high as 25-52.9% (3-8), which is even more than the fatality rate of transplantation rejection (5,9). Among others, intra-abdominal infection (IAI) after LT has obscure symptoms, which is easy to be missed. Meanwhile, its high incidence up to 12.9% to 37.6% (10-13) makes it all the more worthy of attention. In China, most of hepatocellular carcinoma (HCC) patients had a long history of chronic viral hepatitis, partial of them with liver cirrhosis, or with liver function insufficient. Furthermore, HCC grew fast, the nutrition consume increased the burden of the body, and these comprehensive factors can lead to systemic organ function decrease. Thus, the risk factors about postoperative IAI after LT in patients with HCC are fairly complex, it is very necessary to explore.
Materials and methods
General information
A series of 82 patients [74 men (90.2%) and 8 women (9.8%), male:female is 9.25:1] who received orthotopic liver transplantation (OLT) for hepatocellular carcinoma (HCC) in our department between March 2004 and April 2010 was enrolled in this study. Patients aged 28-70 (47.8±9.3) years old. All the patients had a history of hepatitis, including 79 patients of hepatitis B and 3 patients of hepatitis C. 85.4% (70/82) patients had liver cirrhosis, 40.2% (33/82) of them with liver function insufficient (14 patients with Child B and 19 patients with Child C), 8.5% (7/82) of them with preoperative moderate or severe anemia (HGB <90 g/L), 24.4% (20/82) of them with WBC reduction, 32.9% (27/82) of them with low PLT. 11.0% (9/82) of them accompanied with diabetes, and 17.1% (14/82) them had a history of abdominal surgery.
Preoperative preparation
Routine laboratory examination, including blood, urine, dung routine, blood coagulation function, blood grouping, liver function, renal function, hepatitis B/C virus markers (HBVM), tumor markers, abdominal ultrasonography, electrocardiogram (EKG), pulmonary function, chest radiography, gastroscopy upper abdomen CT and MRA.
Operation method
Classic non-bypass (74 cases) or piggyback orthotropic liver transplantation (PBOLT) (8 cases), with additional splenectomy (3 cases) or ligation of splenic artery (2 cases). The donor’s kidney and liver were obtained separately in 12 cases, and together in 70 cases. Mild fatty liver was found in 6 cases, with the others all fat free. Perfusion was performed via the superior mesenteric vein, portal vein trunk or splenic vein intubation, abdominal aortic perfusion fluid: renal liquid 2,000 mL, portal vein: UW solution 4,000 mL, biliary tract: renal liquid 200 mL. The repairing of the donor liver and LT were performed by the same personnel. Vascular anastomosis method: vena cava: hepatic precava and postcava were sutured by continuous everting suture (CES) with 3-0 proline and bloodletting; portal vein by CES with 5-0 proline and bloodletting; artery was trimmed into bell mouth by interrupted suture with 7-0 proline. Bile ducts with the diameter of 4-12 mm were connected by end to end interrupted suture with 7-0 continuous proline without supporting tube. Only 1 case of bilioenteric anastomosis was performed in our group.
Postoperative special care
Postoperative special care was performed to every patient in ICU. Antibiotics were used intra-operation and post-operation at least 3 consecutive days. Postoperative immunosuppression protocol was Steroid+tacrolimus+mycophenolate mofetil (MMF) or basiliximab+tacrolimus+MMF. Postoperative analgesia pump or subcutaneous injection of pethidine hydrochloride for analgesia. Patients accepted gastrointestinal decompression for 2-4 days until anus exhaust. Time to remove the single cavity drainage tube was that abdominal drainage volume was less than 10 mL for 3 consecutive days. Most the patients resumed oral intake within 3-5 days after operation. Blood tests were done postoperative 1, 3, 5, 7, 10, 14 days including blood routine, liver and renal function, electrolyte, blood amylase, abdominal fluid amylase.
Assessment
The judgment standard for diagnosis of early postoperative intra-abdominal infection: abnormal hemogram, temperature more than 38.5 degree centigrade, with blood and liquid culture positive, including bacterial enteritis and fungal enteritis. Diagnosis of postoperative intra-abdominal obvious hemorrhage was according to the total volume of peritoneal blood drainage above 400 mL within the first 72 hours. Postoperative pancreatitis judgment standard was according to the International Study Group of Pancreatic Surgery (ISGPS) definition standard (14): patients with elevated amylase more than twice the upper limit of normal level, without obvious clinical symptoms or typical imaging support were diagnosed as hyperamylasemia (15). Hyoeramylasemia patients with obvious symptoms and pancreatic morphological change or obvious exudation confirmed by ultrasonic gram and CT scan were considered as acute postoperative pancreatitis. Complications occurred within 30 days were considered as the short term postoperative complications. None intra-operative death occurred in this study.
Statistical analysis
SPSS18.0 software was used for statistical analysis. Chi-square test was used in univariate analysis. Logistic regression analysis was used in univariate analysis. P<0.05 was considered statistically significant. According to the group experience and comprehensive reference, 22 items were analysis in this group: (I) preoperative factors: gender, age, abdominal surgery history, diabetes, abdominal cavity effusion, malignant tumor size and grade of Child-Pugh score, hypoalbuminemia, preoperative blood test of white blood cell (WBC), hemoglobin (HGB), platelet (PLT); (II) intraoperative factors: modus operandi, donor fatty liver, cold ischemia time, operation time, anhepatic phase, intraoperative bleeding volume; (III) postoperative factors: immunosuppressive schemes, severe complications (such as postoperative intra-abdominal hemorrhage within the first 72 hours, bile leakage, pancreatitis, etc.).
Results
Bacterial types of infection
A series of 13 patients (13/82, 15.9%) in this group had postoperative IAI. There were 6 patients of G+ bacterial infection including and 11 patients of G- bacterial infection in total, including 4 cases of mixed infection. There were 4 types of G+ infection including 1 case of staphylococcus epidermidis, 2 cases of enterococcus faecalis, 2 cases of staphylococcus aureus and 1 case of staphylococcus haemolyticus. There were 6 types of G- infection including 3 cases of acinetobacter baumannii, 2 cases of pseudomonas aeruginosa, 1 case of maltophilia aeromonas, 2 cases of klebsiella, 1 case of enterobacter cloacae and 2 cases of escherichia coli.
Independent risk factors of postoperative IAI
Univariate analysis suggested postoperative IAI rate was obviously higher in patients with preoperative moderate or severe anemia (HGB <90 g/L) than the remaining patients, 57.1% (4/7) vs. 12.0% (9/75) (P=0.002). And postoperative IAI rate was also increased significantly in patients with obvious postoperative intra-abdominal hemorrhage (>400 mL within the first 72 hours), 50.0% (4/8) vs. 12.2% (9/74) (P=0.005). The remaining factors, such as preoperative Child-Pugh grade, preoperative WBC grade, diameter of tumor, liver cirrhosis, modus operandi, intra-operative bleeding volume, postoperative bile leakage and pancreatitis etc., had no significant correlation with IAI. (Univariate analysis of risk factors for postoperative intra-abdominal infection was shown in Table 1).
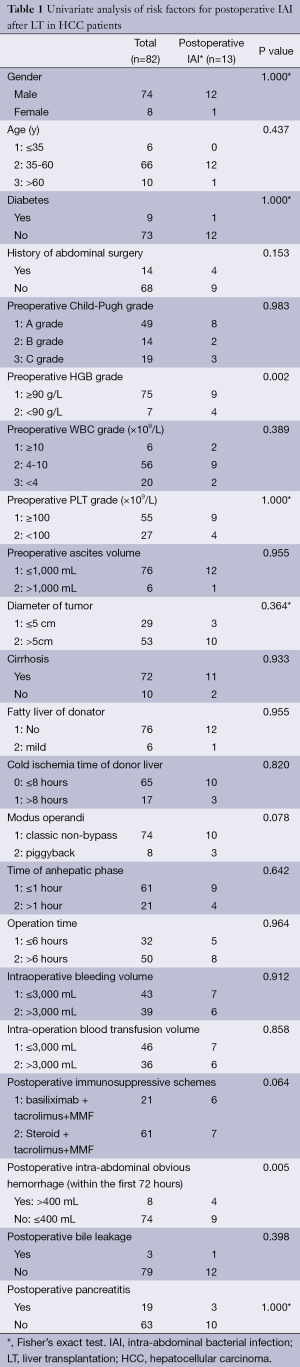
Full table
Sequential analysis of multiple factors suggested that preoperative anemia (HGB <90 g/L), postoperative intra-abdominal hemorrhage (>400 mL within the first 72 hours) were the independent risk factors of postoperative IAI after LT in these HCC patients with the odds ratios at 8.121 (95% CI, 1.417 to 46.550, P=0.019) and 5.911 (95% CI, 1.112 to 31.432, P=0.037). (Multivariate analysis of risk factors for postoperative intra-abdominal infection was shown in Table 2).
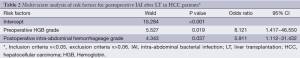
Full table
Discussion
Preoperative moderate or severe anemia may cause postoperative IAI through intestinal flora movement
Preoperative moderate or severe anemia was one of the important risk factors for post-LT IAI in these HCC patients. The IAI rate was obviously increased in patient with moderate or severe anemia (57.1% vs. 12%, P=0.002), according to the remainder patients. Further multi-factor analysis showed that preoperative anemia (HGB <90 g/L) was a independent risk factor for postoperative IAI (P=0.019, OR: 8.121, 95% CI, 1.417 to 46.550). Postoperative IAI is a common complication after LT, in which bacterial infection forms the majority. The main reason of infection is impaired immune function. HCC grew fast, the nutrition consume increased the burden of the body and damaged the body’ immune system. The HCC patients with moderate or severe anemia were susceptible to infection as long term nutrition insufficient, poor body condition, low immune system. In addition, moderate or severe anemia can cause the body’s oxygen supply shortage. Low oxygen environment is easy to cause inflammation, edema, and lead to intestinal mucosal barrier dysfunction, intestinal flora movement. In this article, there were 2 cases of enterococcus faecalis infection, 2 cases of pseudomonas aeruginosa infection, 1 case of enterobacter cloacae and 2 cases of escherichia coli infection. These four types of bacterial are all the intestinal opportunistic pathogens. Therefore, before undergoing LT with HCC patients, we should diagnose and treat anemia actively to avoid postoperative IAI.
Postoperative intra-abdominal hemorrhage formed a good medium for bacteria
Postoperative intra-abdominal obvious hemorrhage was another independent risk factor for postoperative IAI after LT in these 82 HCC patients. Due to the complexity, long time of abdominal opening of liver transplant, the abdominal organs exposed for a long time, which may pose a potential pollution. Abdominal bleeding also is a common complication after LT, with 7.0% incidence according to literature report (16). Long time of abdominal exposure caused the intestinal flora displacement, and the postoperative intra-abdominal hemorrhage or blood clot formed a good medium for pathogenic bacteria. The IAI rate was significantly increased by the above reasons. In our study postoperative IAI rate was 50% (4/8) in patients with postoperative intra-abdominal obvious hemorrhage (>400 mL within the first 72 hours), on the contrary, the IAI rate in patient with bleeding under 400 mL was just 12.2% (9/74) (P=0.005). Multiple factors sequential analysis showed postoperative intra-abdominal obvious hemorrhage (72 h >400 mL) was an independent risk factor of postoperative IAI [5.911 (95% CI, 1.112 to 31.432, P=0.037)]. The common causes of abdominal bleeding after LT included reduced blood coagulation factor in end-stage liver disease, incomplete stopping bleeding during operation process and so on. The coagulation derangement was caused of preoperative liver function, hypersplenism, low PLT, coagulation dysfunction, transfusion of large amounts of banked blood, the application of heparin, etc. In this study, all of these HCC patients were associated with chronic viral hepatitis, 85.4% (70/82) of them with liver cirrhosis, 40.2% (33/82) of patients with liver function insufficient, 32.9% (27/82) of them with PLT decrease. 43.9% (36/82) of them with intraoperative blood transfusion volume larger than 3000ml. Thus, before graft work properly, these negative factors made the patients must undergo a poor coagulation period in the early stage after LT. Small not repaired factors can cause postoperative intra-abdominal hemorrhage. Common bleeding sites were small branches of the vena cava vein (Short hepatic vein, SHV), hepatic artery anastomosis, liver biopsy site, etc. Therefore, to avoid postoperative hemorrhage, it is necessary for preoperative improvement of blood coagulation, PLT and cryoprecipitate supplements, prothrombin complex and fibrinogen, etc. And operators should be careful in the operation procedure, to reduce the incidence of postoperative intra-abdominal hemorrhage. Postoperative ultrasound or CT scanning is helpful to the early detection of abdominal bleeding (17,18). Active bleeding should be treated by medication therapy or surgery. Blood clot should also be remove by surgery, thus to reduce the incidence of intra-abdominal infection.
Postoperative bile leakage and IAI
Postoperative bile leakage was usually considered as a high risk factor for IAI after LT. In this study, the incidence of bile leakage was 3.7%. Oconner et al. (19) reported that the incidence of bile leakage after LT is 5.5-27.4%. Bile chemical stimulation of the abdominal cavity, is easy to cause inflammation, edema, and lead to intestinal mucosal barrier dysfunction, intestinal flora movement. Meanwhile, the reduced biliary pressure during bile leakage could let the intestinal bacteria enter the abdominal cavity by the Oddi’s sphincter. All of the above make facilitation of IAI. But in our study the single factor analysis suggested that postoperative bile leakage wasn’t significantly associated with IAI after LT in HCC patients (P=0.398), and it needs to be confirmed by a larger sample cases. Anyway, as far as bile leakage happened, unobstructed drainage and etiological therapy are necessary for preventing chemical peritonitis and IAI (20).
Above all, IAI is a common early complication after LT in HCC patients. Preoperative anemia (HGB <90 g/L), and postoperative intra-abdominal obvious hemorrhage (72 h >400 mL) are independent risk factors of IAI after LT in HCC patients.
Our study suggested that in order to reduce the occurrence of post-LT IAI in HCC patients, it is necessary to improve anemia before the operation, and stanch bleeding as carefully as possible during the procedure to reduce the postoperative intra-abdominal hemorrhage.
Acknowledgements
This study was funded by a new round of the Shanghai Health System outstanding young talent training plan (XYQ2011030).
Disclosure: The authors declare no conflict of interest.
References
- Lee SG. Current status of liver transplantation in Korea. Korean J Gastroenterol 2005;46:75-83. [PubMed]
- Piselli P, Zanfi C, Corazza V, et al. Incidence and timing of infections after liver transplant in Italy. Transplant Proc 2007;39:1950-2. [PubMed]
- Singh N, Wagener MM, Obman A, et al. Bacteremias in liver transplant recipients: shift toward gram-negative bacteria as predominant pathogens. Liver Transpl 2004;10:844-9. [PubMed]
- Shi SH, Kong HS, Jia CK, et al. Risk factors for pneumonia caused by multidrug-resistant Gram-negative bacilli among liver recipients. Clin Transplant 2010;24:758-65. [PubMed]
- Kim YJ, Kim SI, Wie SH, et al. Infectious complications in living-donor liver transplant recipients: a 9-year single-center experience. Transpl Infect Dis 2008;10:316-24. [PubMed]
- Bedini A, Codeluppi M, Cocchi S, et al. Gram-positive bloodstream infections in liver transplant recipients: incidence, risk factors, and impact on survival. Transplant Proc 2007;39:1947-9. [PubMed]
- Imanieh MH, Erjaee A, Dehghani SM, et al. Early postoperative complications of pediatric liver transplantation. Indian Pediatr 2009;46:1088-90. [PubMed]
- Shi SH, Kong HS, Xu J, et al. Multidrug resistant gram-negative bacilli as predominant bacteremic pathogens in liver transplant recipients. Transpl Infect Dis 2009;11:405-12. [PubMed]
- Shepherd RW, Turmelle Y, Nadler M, et al. Risk factors for rejection and infection in pediatric liver transplantation. Am J Transplant 2008;8:396-403. [PubMed]
- Hashimoto M, Sugawara Y, Tamura S, et al. Pseudomonas aeruginosa infection after living-donor liver transplantation in adults. Transpl Infect Dis 2009;11:11-9. [PubMed]
- Bellier C, Bert F, Durand F, et al. Risk factors for Enterobacteriaceae bacteremia after liver transplantation. Transpl Int 2008;21:755-63. [PubMed]
- Hashimoto M, Sugawara Y, Tamura S, et al. Bloodstream infection after living donor liver transplantation. Scand J Infect Dis 2008;40:509-16. [PubMed]
- Reid GE, Grim SA, Sankary H, et al. Early intra-abdominal infections associated with orthotopic liver transplantation. Transplantation 2009;87:1706-11. [PubMed]
- Wente MN, Veit JA, Bassi C, et al. Postpancreatectomy hemorrhage (PPH): an International Study Group of Pancreatic Surgery (ISGPS) definition. Surgery 2007;142:20-5. [PubMed]
- Alexander JA, Demetrius AJ, Gavaler JS, et al. Pancreatitis following liver transplantation. Transplantation 1988;45:1062-5. [PubMed]
- Wu YM, Voigt M, Rayhill S, et al. Suprahepatic venacavaplasty (cavaplasty) with retrohepatic cava extension in liver transplantation: experience with first 115 cases. Transplantation 2001;72:1389-94. [PubMed]
- Boraschi P, Donati F. Complications of orthotopic liver transplantation: imaging findings. Abdom Imaging 2004;29:189-202. [PubMed]
- Pandharipande PV, Lee VS, Morgan GR, et al. Vascular and extravascular complications of liver transplantation: comprehensive evaluation with three-dimensional contrast-enhanced volumetric MR imaging and MR cholangiopancreatography. AJR Am J Roentgenol 2001;177:1101-7. [PubMed]
- O’Connor TP, Lewis WD, Jenkins RL. Biliary tract complications after liver transplantation. Arch Surg 1995;130:312-7. [PubMed]
- Mosca S, Militerno G, Guardascione MA, et al. Late biliary tract complications after orthotopic liver transplantation: diagnostic and therapeutic role of endoscopic retrograde cholangiopancreatography. J Gastroenterol Hepatol 2000;15:654-60. [PubMed]


